

Coming to the Annual Meeting of The American Society of Breast Surgeons?
We hope to see you there at the Medneon booth #353, which will have iGAP Registry on-boarding examples and opportunities to meet our lead investigators!
The American Society of Breast Surgeons' Annual Meeting is April 30th - May 5th, 2019 in Dallas, Texas.
Learn more by visiting the ASBrS Annual Meeting website.
The President of the American Society of Breast Surgeons, Dr. Walton Taylor, addressed members today and announced a change in the Society's recommendations for genetic testing. This change was based on the work by PI Dr. Peter Beitsch demonstrating that current testing guidelines miss about half of patients with a mutation that have clinical management recommendations.
Dr. Taylor wrote: "Too many patients develop cancers that might have been prevented or found earlier if genetic testing had been performed. Our Society has a responsibility to act when we see an opportunity to decrease unnecessary morbidity and mortality. We do so today with the adoption of our new position on this issue."
Read the whole story on The ASCO Post.
The Washington Post featured work done by PI Dr. Peter Beitsch on the Universal Breast Cancer Genetic Testing Registry in their article "Breast surgeons say all breast cancer patients should be offered genetic testing".
Read the full article on The Washington Post.
The genesis of the iGAP Registry was the Universal Breast Cancer Genetic Testing Registry, whose PI was Dr. Peter Beitsch, one of the the PIs of the iGAP Registry. The Universal Registry tested breast cancer patients who did and did not meet requirements for genetic testing at 20 academic and community centers across the United States.
The Universal Breast Cancer Genetic Testing Registry demonstrated that nearly half of patients with breast cancer with a P/LP variant that has clinically actionable and/or management guidelines in development are missed by current testing guidelines.
These results were published in the Journal of Clinical Oncology in December 2018 and were utilized by the American Society of Breast Surgeons to define new testing guidelines which were published in 2019.
The iGAP Registry provides a platform by which community and academic physicians can participate in further research that redefines future testing guidelines and utilization.
Read the full Universal Breast Cancer Genetic Testing Registry JCO publication here.
The Universal Breast Cancer Genetic Testing Registry laid the groundwork for the iGAP Registry in understanding the ability of the NCCN guidelines to identify patients with P/LP mutations.
This poster reviews the 949 subjects analyzed, of which 49.95% met NCCN criteria for genetic testing and 50.05% did not meet criteria. Demographics and P/LP Variants by NCCN criteria are compared.
These data demonstrate that patients who met NCCN genetic testing guidelines have similar likelihood of harboring a variant compared to those who do not.
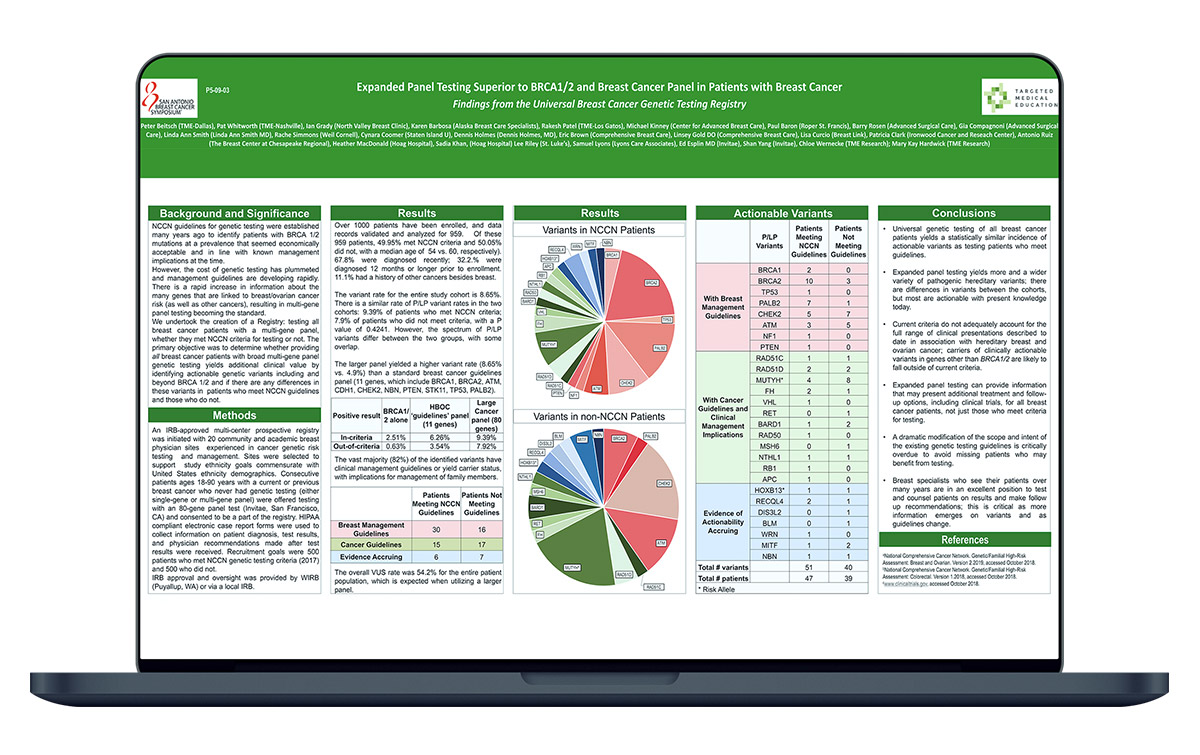
The Universal Breast Cancer Genetic Registry sought to determine whether providing all breast cancer patients with broad multi-gene panel genetic testing yields additional clinical value and if there was a difference in variants in patients who met NCCN guidelines and those who did not.
Expanded panel testing yields more and a wider variety of pathogenic hereditary variants; there are differences in variants between the cohorts, but most are actionable with present knowledge today. Current criteria do not adequately account for the full range of clinical presentations described to date in association with hereditary breast and ovarian cancer; carriers of clinically actionable variants in genes other than BRCA 1/2 are likely to fall outside of current criteria.